Lithium-ion batteries have long dominated the market as the go-to power source for electric vehicles. They are also increasingly being considered for storage of renewable energy to be used on the electric grid. However, with the rapid expansion of this market, supply shortages of lithium are projected within the next five to 10 years.
“Sodium-ion batteries are emerging as a compelling alternative to lithium-ion batteries due to the greater abundance and lower cost of sodium,” said Gui-Liang Xu, a chemist at the U.S. Department of Energy’s (DOE) Argonne National Laboratory.
To date, there has been a serious roadblock to commercialization of such batteries. In particular, the performance of the sodium-containing cathode rapidly declines with repeated discharge and charge.
A team at Argonne has made important strides in resolving this issue with a new design for a sodium-ion oxide cathode. It is closely based on an earlier Argonne design for a lithium-ion oxide cathode with proven high energy storage capacity and long life.
A key feature of both designs is that the microscopic cathode particles contain a mix of transition metals, which could include nickel, cobalt, iron or manganese. Importantly, these metals are not uniformly distributed in individual cathode particles. As an example, nickel appears at the core; surrounding this core are cobalt and manganese, which form a shell. These elements serve different purposes. The manganese-rich surface gives the particle its structural stability during charge-discharge cycling. The nickel-rich core provides high capacity for energy storage.
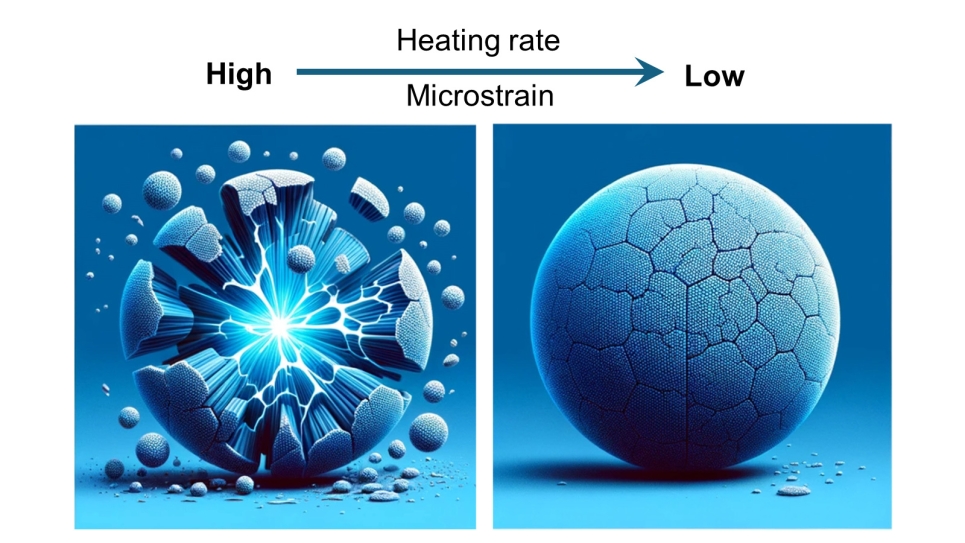
In testing this design, however, the cathode’s energy storage capacity steadily declined during cycling. The problem was traced to the formation of cracks in the particles during cycling. These cracks formed due to strain arising between the shell and core in the particles. The team sought to eliminate that strain before cycling by fine-tuning their method of cathode preparation.
The precursor material used to start the synthesis process is a hydroxide. In addition to oxygen and hydrogen, it contains three metals: nickel, cobalt and manganese. The team made two versions of this hydroxide: One with the metals distributed in a gradient from core to shell and, for comparison, another with the three metals evenly distributed throughout each particle.
To form the final product, the team heated up a mixture of a precursor material and sodium hydroxide to as high as 600 degrees Celsius, maintained it at that temperature for a select period, then cooled it to room temperature. They also tried different heat-up rates.
During this entire treatment, the team monitored the structural changes in the particle properties. This analysis involved use of two DOE Office of Science user facilities: the Advanced Photon Source (beamlines 17-BM and 11-ID) at Argonne and the National Synchrotron Light Source II (beamline 18-ID) at DOE’s Brookhaven National Laboratory.
“With the X-ray beams at these facilities, we could determine real-time changes in the particle composition and structure under realistic synthesis conditions,” said Argonne beamline scientist Wenqian Xu.
The team also used the Center for Nanoscale Materials (CNM) at Argonne for additional analysis to characterize the particles and the Polaris supercomputer at the Argonne Leadership Computing Facility (ALCF) to reconstruct the X-ray data into detailed 3D images. The CNM and ALCF are also DOE Office of Science user facilities.
The initial results revealed no cracks in the uniform particles, but cracks forming in the gradient particles at temperatures as low as 250 degrees C. These cracks appeared at the core and the core-shell boundary and then moved to the surface. Clearly, the metal gradient caused significant strain leading to these cracks.
“Since we know that gradient particles can produce cathodes with high energy storage capacity, we wanted to find heat treatment conditions that will eliminate the cracks in the gradient particles,” said Wenhua Zuo, an Argonne postdoctoral appointee.
The heat-up rate proved a critical factor. Cracks formed at a heat-up rate of five degrees per minute, but not at a slower rate of one degree per minute. Tests in small cells with cathode particles prepared at the slower rate maintained their high performance for over 400 cycles.
“Preventing cracks during cathode synthesis pays big dividends when the cathode is later charged and discharged,” said Gui-Liang Xu. “And while sodium-ion batteries do not yet have sufficient energy density to power vehicles over long distances, they are ideal for urban driving.”
The team is now working to eliminate the nickel from the cathode, which would reduce the cost even further and be more sustainable.
“The prospects seem very good for future sodium-ion batteries with not only low cost and long life, but also energy density comparable to that of the lithium iron phosphate cathode now in many lithium-ion batteries,” said Khalil Amine, an Argonne Distinguished Fellow. “This would result in more sustainable electric vehicles with good driving range.”
Funding was provided by the DOE Vehicle Technologies Office, Office of Energy Efficiency and Renewable Energy, and Advanced Scientific Computing Research Program.
This research first appeared in Nature Nanotechnology. In addition to Zuo, Wenqian Xu, Gui-Liang Xu and Amine, Argonne authors include Jihyeon Gim, Tianyi Li, Dewen Hou, Yibo Gao, Shiyuan Zhou, Chen Zhao, Xin Jia, Zhenzhen Yang and Yuzi Liu. Also contributing was Xianghui Xiao from Brookhaven.
About Argonne’s Center for Nanoscale Materials
The Center for Nanoscale Materials is one of the five DOE Nanoscale Science Research Centers, premier national user facilities for interdisciplinary research at the nanoscale supported by the DOE Office of Science. Together the NSRCs comprise a suite of complementary facilities that provide researchers with state-of-the-art capabilities to fabricate, process, characterize and model nanoscale materials, and constitute the largest infrastructure investment of the National Nanotechnology Initiative. The NSRCs are located at DOE’s Argonne, Brookhaven, Lawrence Berkeley, Oak Ridge, Sandia and Los Alamos National Laboratories. For more information about the DOE NSRCs, please visit https://science.osti.gov/User-Facilities/User-Facilities-at-a-Glance.
The Argonne Leadership Computing Facility provides supercomputing capabilities to the scientific and engineering community to advance fundamental discovery and understanding in a broad range of disciplines. Supported by the U.S. Department of Energy’s (DOE’s) Office of Science, Advanced Scientific Computing Research (ASCR) program, the ALCF is one of two DOE Leadership Computing Facilities in the nation dedicated to open science.
About the Advanced Photon Source
The U. S. Department of Energy Office of Science’s Advanced Photon Source (APS) at Argonne National Laboratory is one of the world’s most productive X-ray light source facilities. The APS provides high-brightness X-ray beams to a diverse community of researchers in materials science, chemistry, condensed matter physics, the life and environmental sciences, and applied research. These X-rays are ideally suited for explorations of materials and biological structures; elemental distribution; chemical, magnetic, electronic states; and a wide range of technologically important engineering systems from batteries to fuel injector sprays, all of which are the foundations of our nation’s economic, technological, and physical well-being. Each year, more than 5,000 researchers use the APS to produce over 2,000 publications detailing impactful discoveries, and solve more vital biological protein structures than users of any other X-ray light source research facility. APS scientists and engineers innovate technology that is at the heart of advancing accelerator and light-source operations. This includes the insertion devices that produce extreme-brightness X-rays prized by researchers, lenses that focus the X-rays down to a few nanometers, instrumentation that maximizes the way the X-rays interact with samples being studied, and software that gathers and manages the massive quantity of data resulting from discovery research at the APS.
This research used resources of the Advanced Photon Source, a U.S. DOE Office of Science User Facility operated for the DOE Office of Science by Argonne National Laboratory under Contract No. DE-AC02-06CH11357.
Argonne National Laboratory seeks solutions to pressing national problems in science and technology by conducting leading-edge basic and applied research in virtually every scientific discipline. Argonne is managed by UChicago Argonne, LLC for the U.S. Department of Energy’s Office of Science.
The U.S. Department of Energy’s Office of Science is the single largest supporter of basic research in the physical sciences in the United States and is working to address some of the most pressing challenges of our time. For more information, visit https://energy.gov/science.